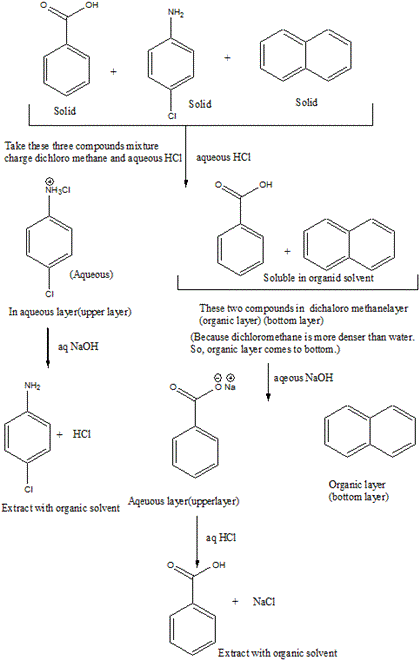
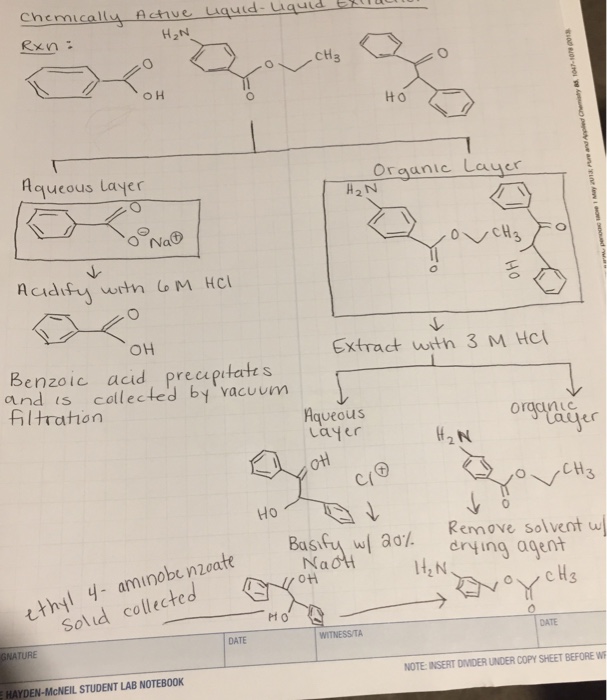
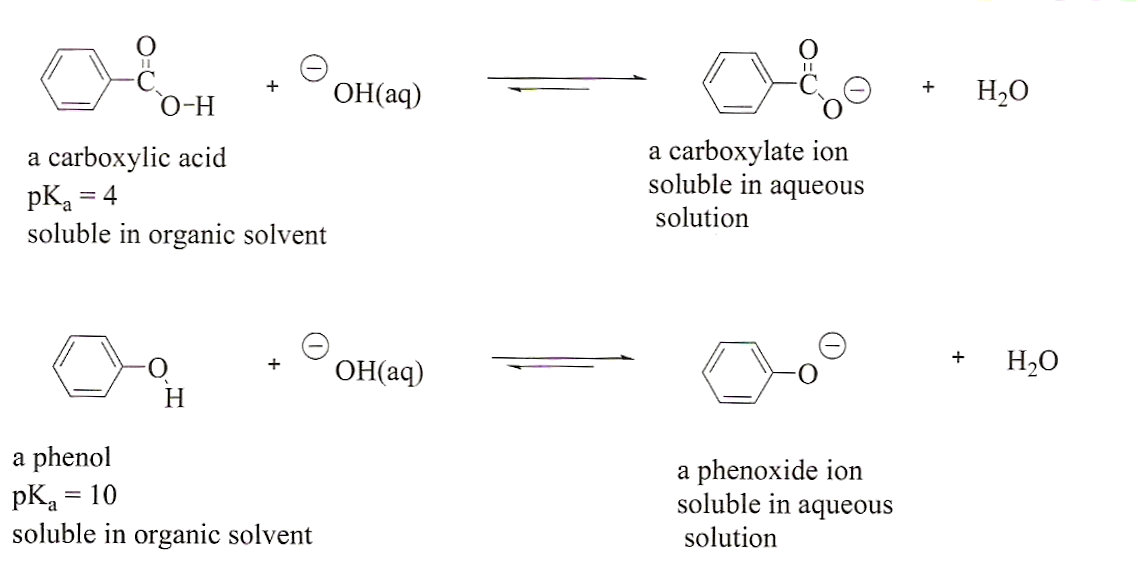
A mixture contains benzoic acid, 4-chloroaniline, and naphthalene. The 4-chloroaniline is separated first by extraction with hydrochloric acid. Since no phenolic compound is present in this mixture, t | Homework.Study.com
Experiment 3: Separation of a Mixture by Acid-Base Extraction – Department of Chemistry – UW–Madison
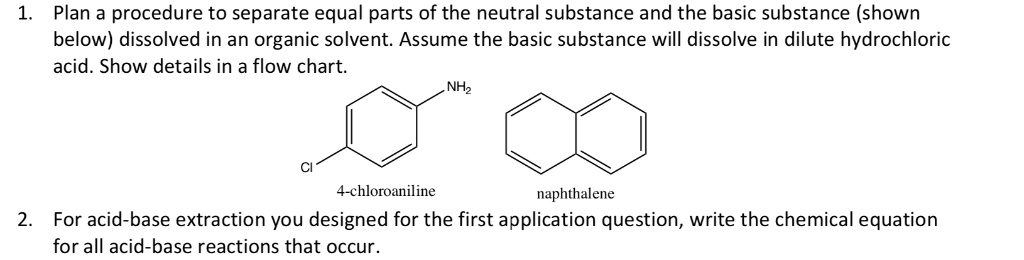
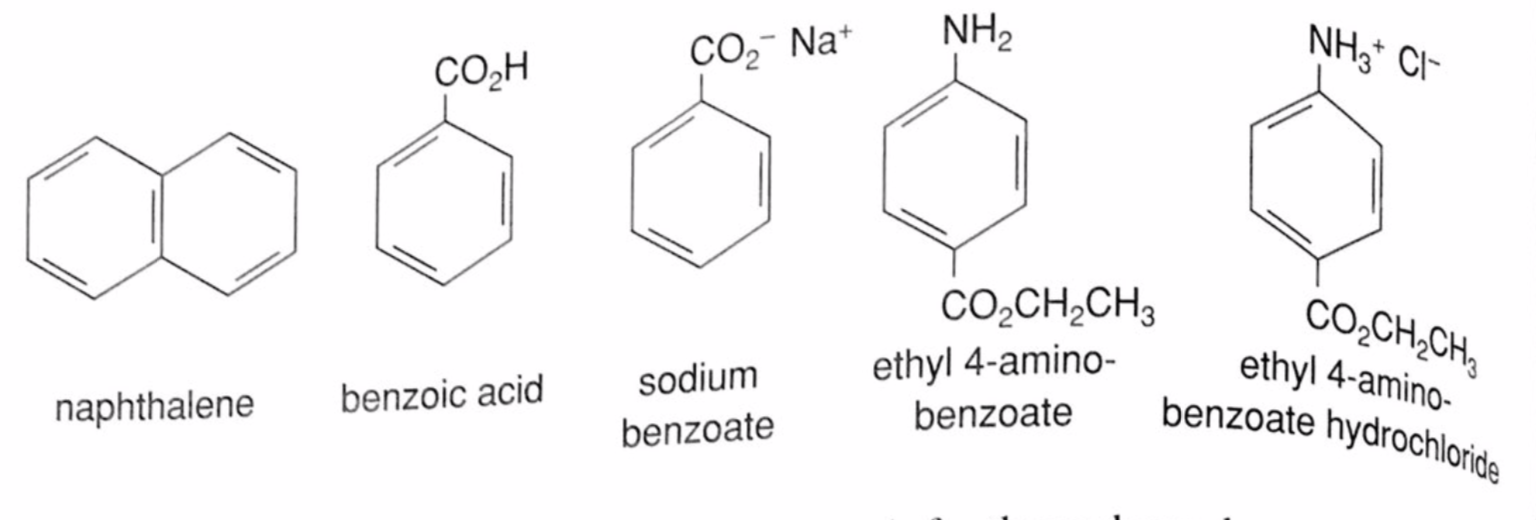
SOLVED: Plan a procedure to separate equal parts of the neutral substance and the basic substance (shown below) dissolved in an organic solvent: Assume the basic substance will dissolve in dilute hydrochloric
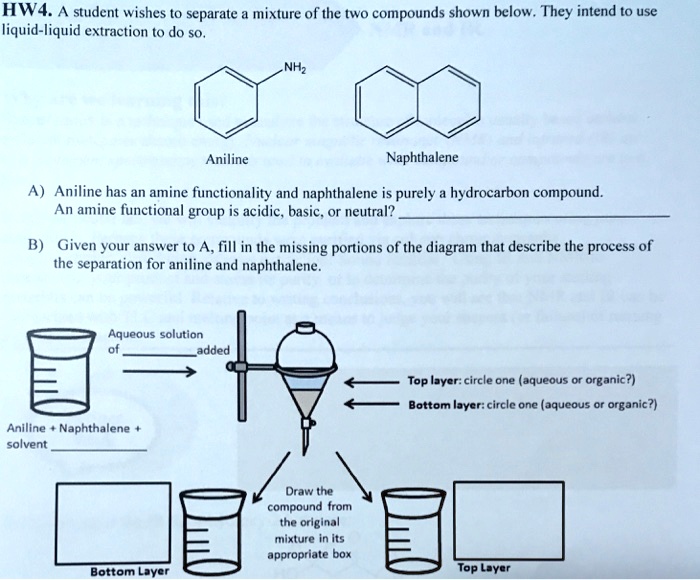
SOLVED: HW4 A student wishes t0 separate mixture of the two compounds shown below They intend t0 use liquid-liquid extraction t0 do so Aniline Naphthalene Aniline has an amine fiunctionality and naphthalene

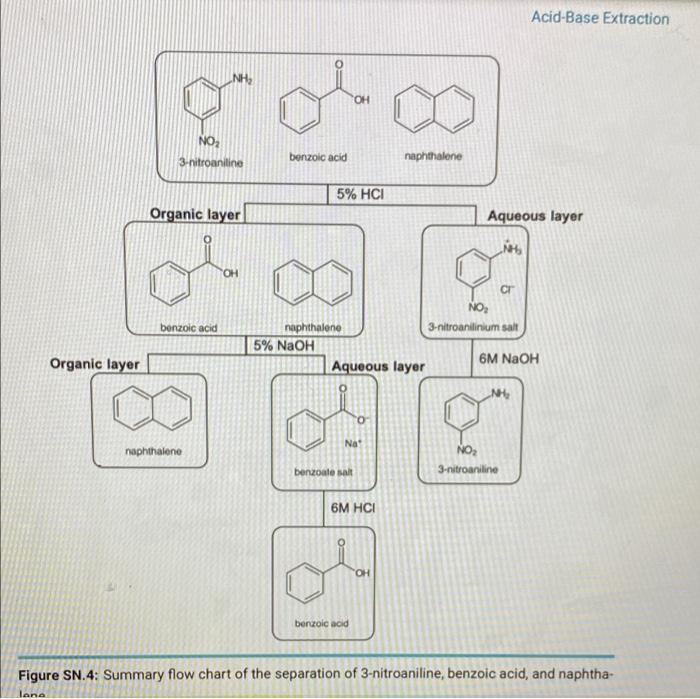
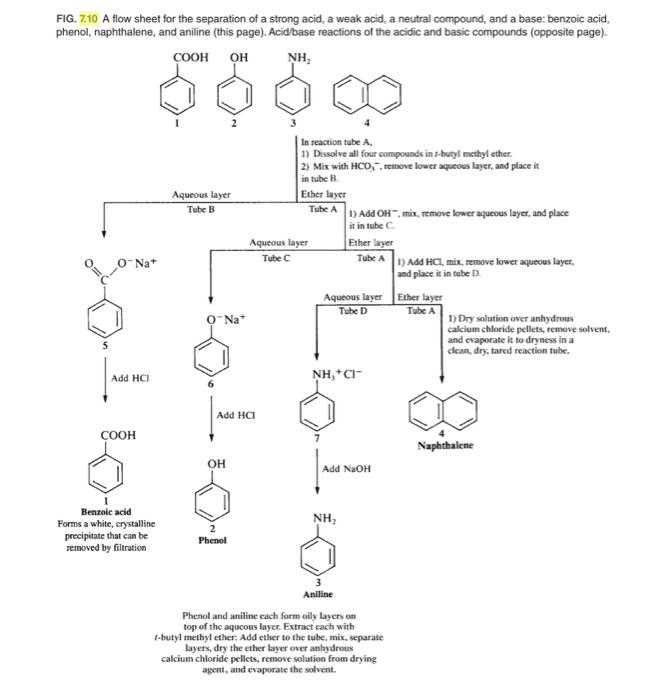
Write out a detailed flow scheme for the separation of naphthalene, benzoic acid and aniline. Use H_2O, HCl, NaOH and methylene chloride reagent/solvents for this. Note that benzoic acid and aniline forms
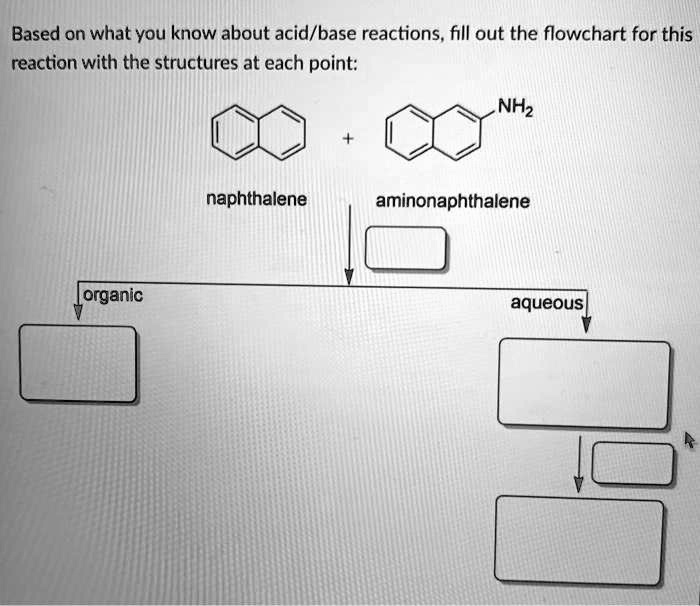
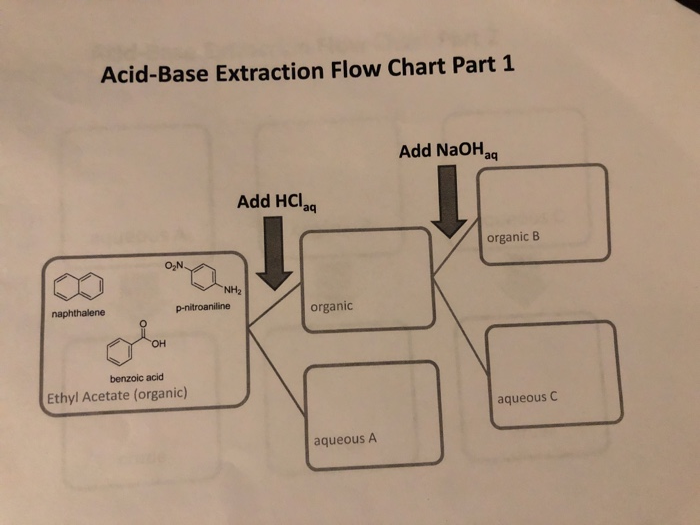
SOLVED: Based on what you know about acid/base reactions, fill out the flowchart for this reaction with the structures at each point: NHz naphthalene aminonaphthalene [organic aqueous
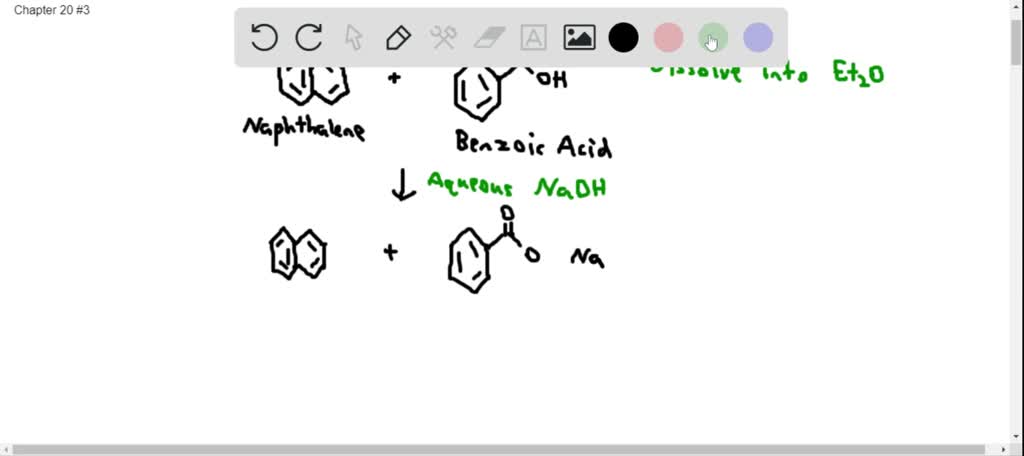
SOLVED:Assume you have a mixture of naphthalene and benzoic acid that you want to separate. How might you take advantage of the acidity of one component in the mixture to effect a