New Drug Therapy for Kidney Stones: A Review of Cellulose Sodium Phosphate, Acetohydroxamic Acid, and Potassium Citrate

Resin-supported arylstannanes as precursors for radiolabeling with iodine: benzaldehydes, benzoic acids, benzamides, and NHS esters
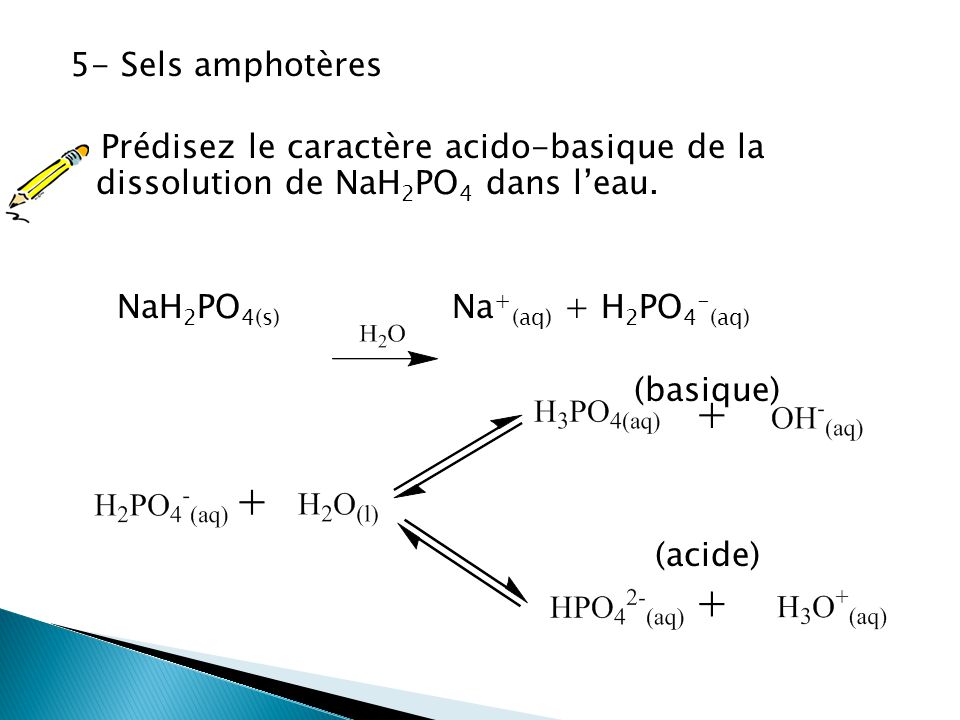
G) Sels Dans le chapitre 1, Acide + Base Sel + Eau HCl(aq) + NaOH(aq) NaCl(aq) + H2O Les sels sont des électrolytes forts qui se dissocient entièrement. - ppt video online télécharger

Analytical ultracentrifugation analyses of the complex SEPT6-C/SEPT7-C... | Download Scientific Diagram

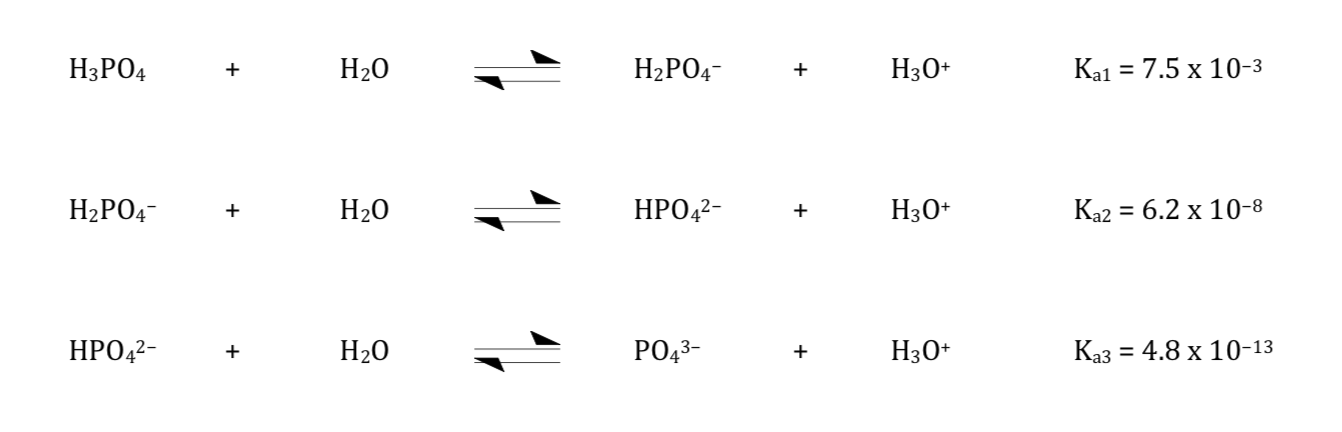
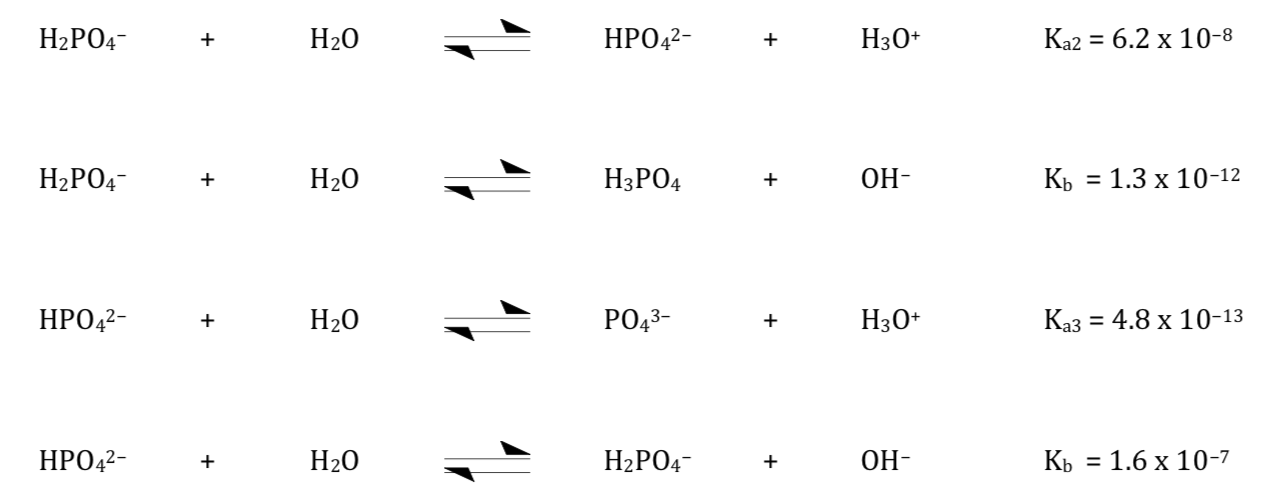
Aqueous solutions of NMA, Na2HPO4, and NaH2PO4 as models for interaction studies in phosphate–protein systems - ScienceDirect

Palladium-Catalyzed Deracemization of Allylic Carbonates in Water with Formation of Allylic Alcohols: Hydrogen Carbonate Ion as Nucleophile in the Palladium-Catalyzed Allylic Substitution and Kinetic Resolution | Journal of the American Chemical Society

WO2015025111A1 - Method for the industrial production of flour from lipid-rich microalga biomass with no "off-notes" by controlling the oxygen availability - Google Patents