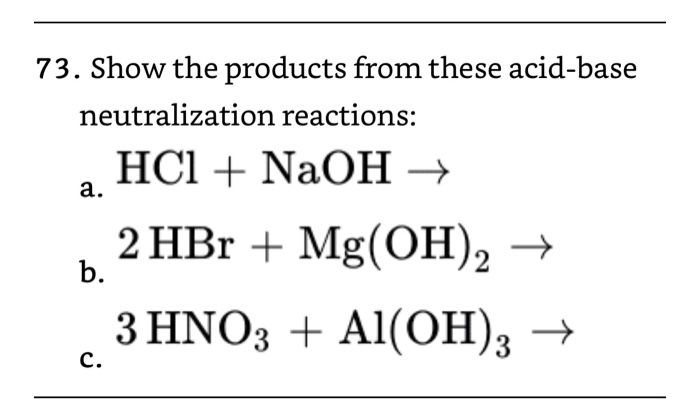
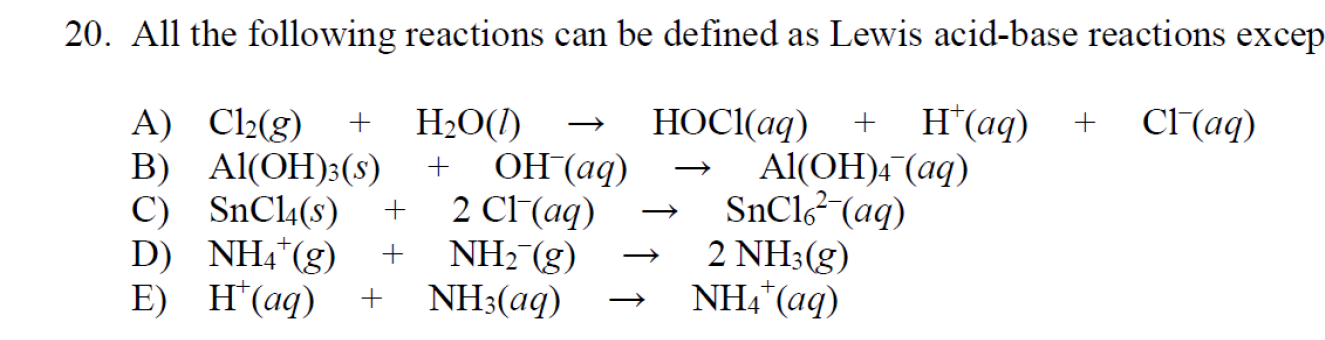Acids and Bases - the Three Definitions 1.Measurement of pH - the pH meter 2.Bronsted-Lowry definition of acids and bases - an acid is a proton donor - - ppt download

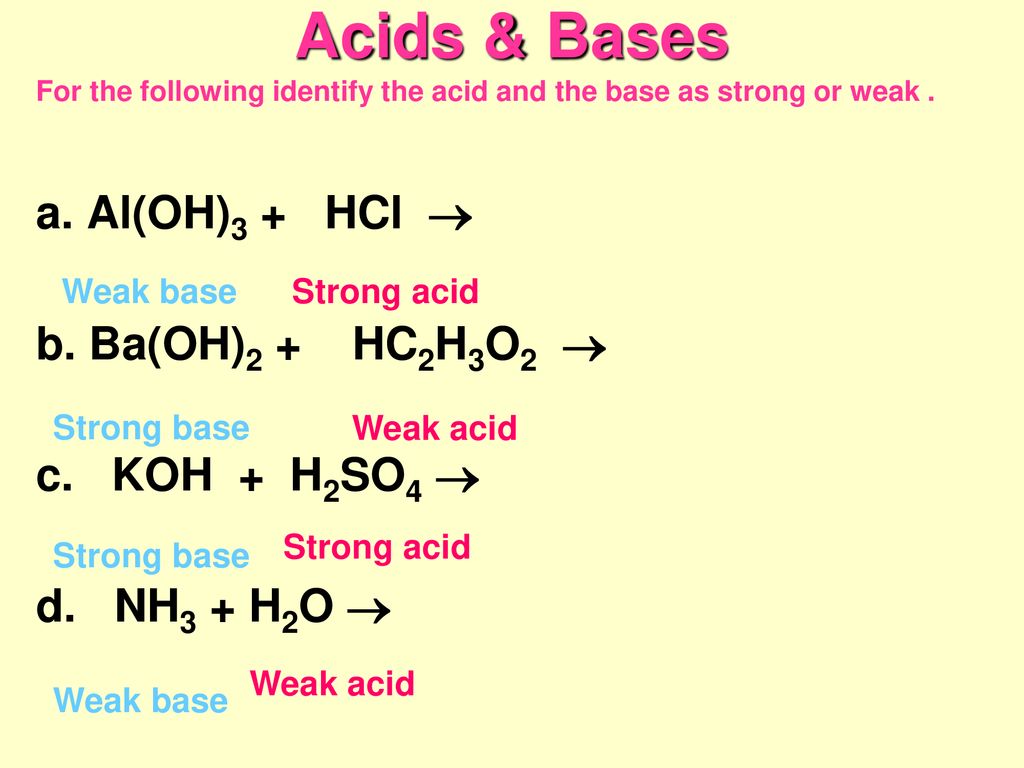
Acid Base Definitions Originally recognized by properties like taste, feel, reactions with indicators – Acids taste sour and turn blue litmus red – Bases. - ppt download
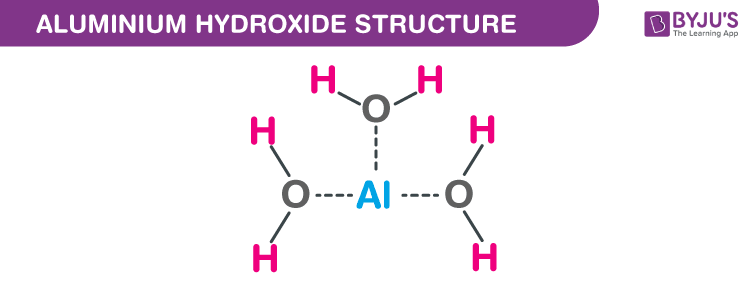
Aluminium Hydroxide (Al(OH)<sub>3</sub>) - Structure, Molecular mass, Properties, Uses and FAQs of Aluminium Hydroxide

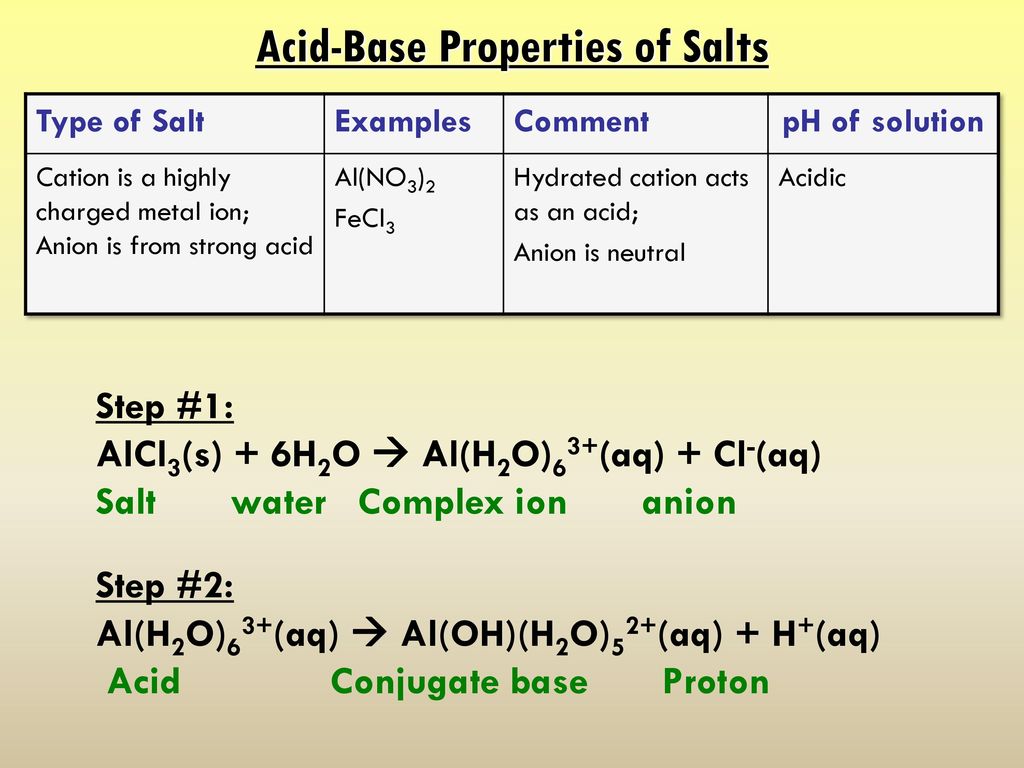
SOLVED: Predict the products of each acid-base combination listed. Assume that a neutralization reaction occurs. HBr and Fe(OH)3 HNO2 and Al(OH)3 HClO3 and Mg(OH)2


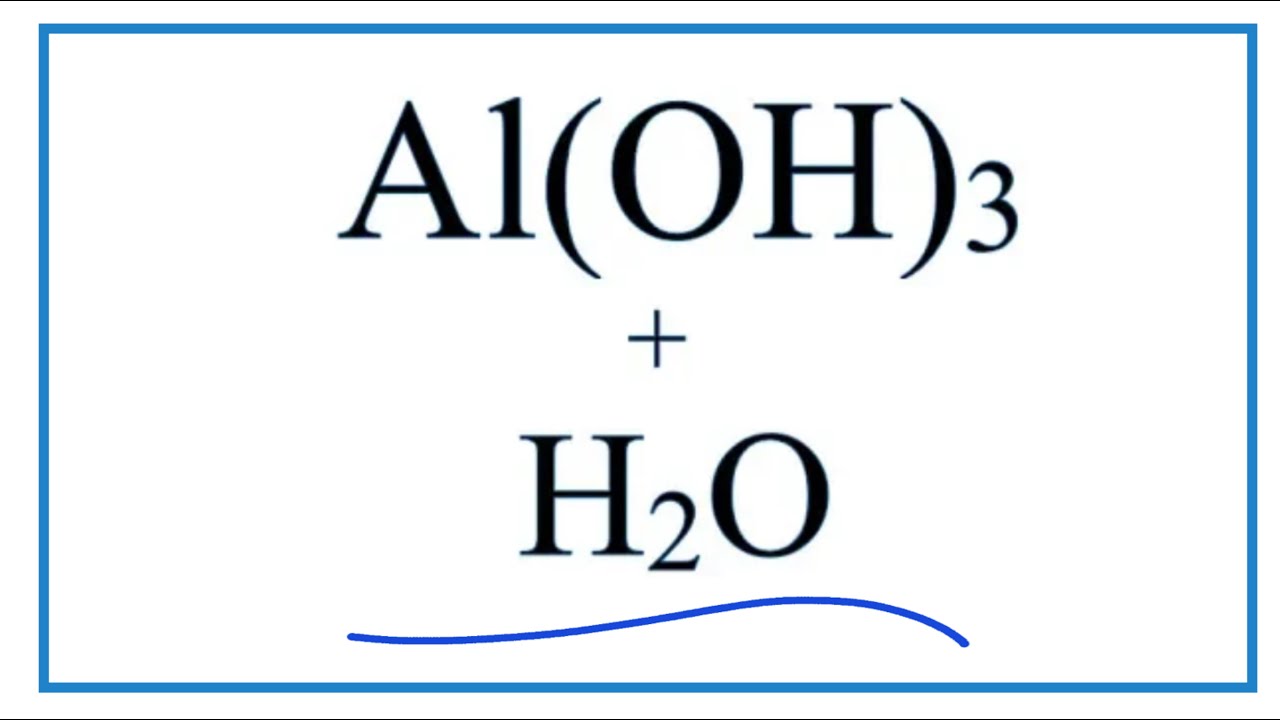
![Solved 7. a) [6pts] Complete and balance the following | Chegg.com Solved 7. a) [6pts] Complete and balance the following | Chegg.com](https://media.cheggcdn.com/study/923/923dd385-3584-435f-942a-aadd87ea16a1/image.png)